Abstract
Background: The post-HERCULES trial (NCT02878603) evaluated long-term safety and efficacy of caplacizumab in patients with acquired thrombotic thrombocytopenic purpura (aTTP; also known as immune-mediated TTP), and efficacy of repeated use of caplacizumab for aTTP recurrence.
Methods: Patients who completed the HERCULES trial were invited to participate in the post-HERCULES study and attend twice-yearly visits for 3 years. In case of aTTP recurrence, patients could receive open-label (OL) caplacizumab with therapeutic plasma exchange (TPE) and immunosuppression (IST). Safety was assessed during the overall study period (intention-to-observe [ITO] population) and during recurrences. TTP-related events (recurrence, mortality, or major thromboembolic events) were assessed in the efficacy ITO population. The ITO population (n=104) included all enrolled patients, grouped by whether they received caplacizumab during HERCULES (randomized or switched to OL after exacerbation). The efficacy ITO population (n=78) included those without aTTP recurrence during HERCULES or prior to the beginning of post-HERCULES. The recurrence population (n=19) included patients with ≥1 recurrence during post-HERCULES; the repeat-use population (n=9) was a subset treated at least twice with caplacizumab (received caplacizumab in HERCULES or treated twice in post-HERCULES). Pharmacodynamics and immunogenicity were assessed in the ITO population. The study was conducted in accordance with the Declaration of Helsinki.
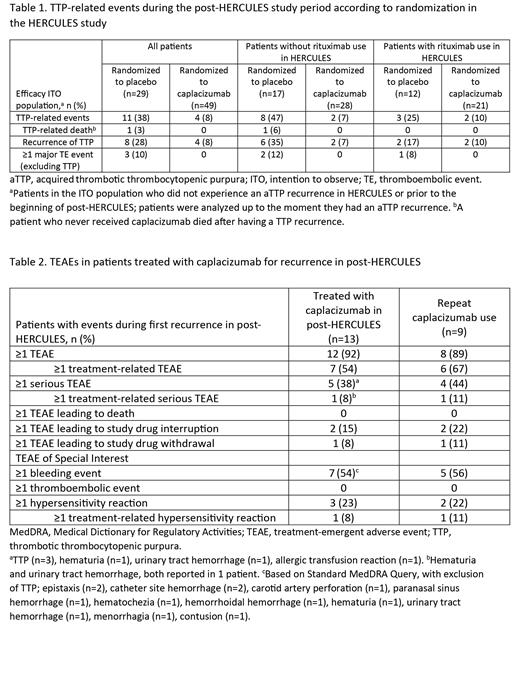
Results: Of 104 patients enrolled in post-HERCULES, 75 were treated with caplacizumab along with TPE+IST during HERCULES (caplacizumab group) and 29 were treated with TPE+IST only (placebo group). During the overall post-HERCULES study period, incidence of adverse events (AEs) and serious AEs was similar between the groups. In the ITO population, recurrence occurred in 11/75 patients (15%) in the caplacizumab group and 8/29 patients (28%) in the placebo group. In the efficacy ITO population, TTP-related events occurred in 8% of patients (4/49) randomized to caplacizumab versus 38% (11/29) randomized to placebo (Table 1). Prior rituximab use during HERCULES was similar in both groups (21/49 patients [43%] randomized to caplacizumab and 12/29 [41%] randomized to placebo). Incidence of recurrence by prior rituximab use (with vs without) was 10% (2/21) versus 7% (2/28) among patients randomized to caplacizumab, and 17% (2/12) versus 35% (6/17) among patients randomized to placebo. Of 19 patients with ≥1 recurrence, 13 were treated with caplacizumab and 6/13 received concomitant rituximab. Recurrences were resolved (12/13) or resolving (1/13) for all patients treated with caplacizumab, including 9 patients with repeat caplacizumab use. One patient who did not receive caplacizumab in either HERCULES or post-HERCULES died as an outcome of recurrence. Safety profile of caplacizumab for treatment of recurrence was consistent with that observed in HERCULES (Table 2) . The most common treatment-emergent AEs (TEAEs) with caplacizumab during the first recurrence (n=13) were headache and constipation (n=3 each). In the repeat-use population (n=9), bleeding events were reported in 5/9 patients (56%) for the first recurrence; 2 patients had a serious treatment-related TEAE of genitourinary or gastrointestinal bleeding; 2 patients had treatment-emergent anti-drug antibodies (ADAs) positive in a functional neutralizing antibody assay. No apparent impact of treatment-emergent ADAs on RICO activity or change from baseline in von Willebrand factor antigen concentration was found.
Conclusions: The long-term safety profile of patients exposed to caplacizumab together with TPE+IST was similar to those who received IST+TPE only, with no observed increases in recurrence of aTTP. Repeat caplacizumab use was efficacious, with no evidence of safety concerns or boosting of ADA response.
Funding: This research was funded by Ablynx, a Sanofi company.
Scully: Shire: Research Funding; Novartis: Research Funding, Speakers Bureau; Takeda: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Sanofi: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Octopharma: Speakers Bureau. de la Rubia: Ablynx/Sanofi: Consultancy; Amgen: Consultancy, Membership on an entity's Board of Directors or advisory committees; Amgen, Bristol Myers Squibb,: Honoraria, Speakers Bureau; Celgene, Takeda, Janssen, Sanofi: Honoraria; Celgene: Consultancy; Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: TRAVEL, ACCOMMODATIONS, EXPENSES; Sanofi: Membership on an entity's Board of Directors or advisory committees; Takeda: Consultancy; AbbVie: Consultancy; Bristol Myers Squibb: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Travel Accommodations; GSK: Consultancy. Pavenski: Alexion: Honoraria, Research Funding; Bioverativ: Research Funding; Ablynx: Honoraria, Research Funding; Octapharma: Research Funding; Shire: Honoraria. Metjian: Momenta/Johnson & Johnson, Sanofi Aventis, Takeda: Research Funding; Ablynx/Sanofi: Consultancy; Sanofi Aventis, Takeda: Other: Advisory. Knoebl: Ablynx/Sanofi: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Shire/Takeda: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; CSL Behring: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Roche: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Novo Nordisk: Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau. Peyvandi: Roche: Consultancy, Honoraria; Takeda: Consultancy, Honoraria; Sanofi: Consultancy, Honoraria; Sobi: Consultancy, Honoraria. Cataland: Alexion: Consultancy, Research Funding; Ablynx/Sanofi: Consultancy, Research Funding; Takeda: Consultancy; Sanofi Genzyme: Consultancy. Coppo: Alexion Pharmaceuticals, Sanofi Aventis, Octapharma AG: Consultancy, Other: Advisory; Alexion Pharmaceuticals, Sandoz, Sanofi Aventis, Takeda: Other: Speakers Bureau; Ablynx/Sanofi: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Shire: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau. Kremer Hovinga Strebel: Ablynx/Sanofi: Membership on an entity's Board of Directors or advisory committees, Other: Speaker at symposia; Shire: Membership on an entity's Board of Directors or advisory committees, Research Funding; CSL Behring: Other: Speaker at symposia; Roche: Other: Speaker at symposia; Siemens: Other: Speaker at symposia. Minkue Mi Edou: Sanofi: Current Employment, Other: May hold shares and/or stock options. de Passos Sousa: Sanofi: Current Employment, Other: May hold shares and/or stock options. Callewaert: Sanofi: Current Employment, Other: May hold shares and/or stock options. Gunawardena: Sanofi: Current Employment, Other: May hold shares and/or stock options. Lin: Sanofi: Current Employment, Other: May hold shares and/or stock options.
Author notes
 This icon denotes a clinically relevant abstract
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal